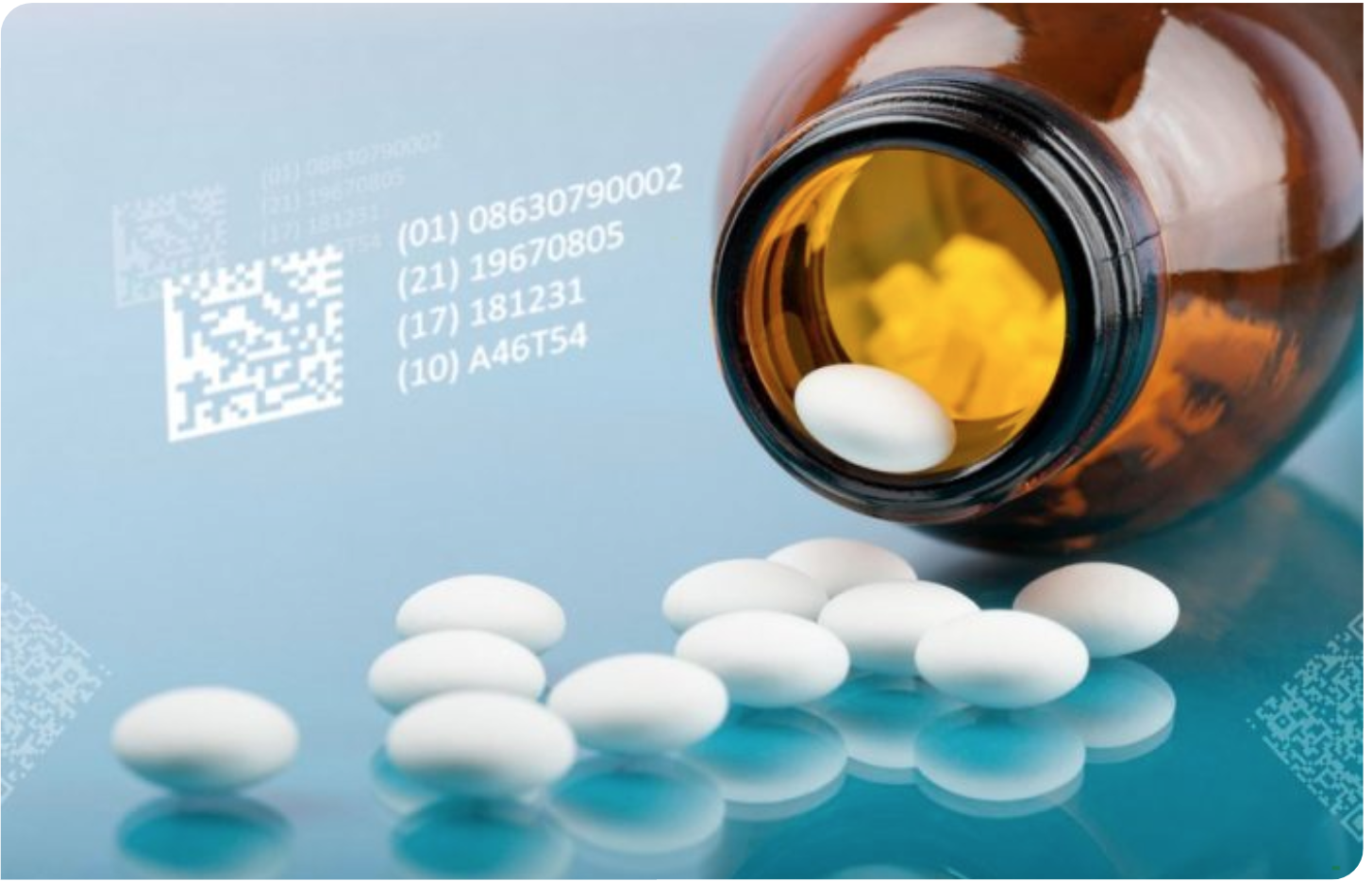
SISTEM SERIALIZACIJE
Ponarejena zdravila predstavljajo tveganje za bolnike. Varnost pacientov in celovitost izdelka sta temeljni vrednoti, zato številne države po svetu izvajajo predpise za boj proti ponarejanju zdravil in zagotavljajo, da so zdravila varna.
Serializacija ali edinstvena identifikacija nam pomaga ugotoviti, kdo je izdelek izdelal, kateri trgovec ga je prodal prodajalcem na drobno ali bolnišnici in kateremu kupcu je bil predpisan. Izvajanje serializacije in sledenja zagotavljata sposobnost sledenja življenjskega cikla zdravil.
Serializacijski sistemi in procesi omogočajo izvajanje postopkov sledenja v vsakem trenutku, ne samo v postopku dobave, temveč tudi med logističnimi in trženjskimi procesi.